2Which Products Does the NMPA Usability Guideline Apply To?
In principle, the NMPA Usability Guideline applies to medical devices of all classes. However, the Guideline requires the submission of usability documentation exclusively for Class II and Class III medical devices. In alignment with IEC 62366‑1:2015, the Guideline does not explicitly state that drug–device combination products fall within its scope of application. Nevertheless, based on various passages of the Guideline—particularly Section VI.(IV)—it can be assumed that the outlined principles also apply to all Class II and Class III medical devices that are part of, or constitute a component of, a product regulated as a drug. In‑vitro diagnostic reagents remain excluded from the scope of application.
It should be noted that the Chinese risk classification is not equivalent to the U.S. or European classification systems. However, this risk classification decisively determines the scope of usability activities to be conducted, in particular with regard to usability testing involving representative users. The following table presents the product catalogue included in the Guideline for products classified as high use‑risk from the perspective of the NMPA, for which summative usability testing is required under certain conditions (see Chapter 3).
- Cardiac radio-frequency ablation equipment
- Cardiac radio-frequency ablation catheter
- Radio-frequency ablation equipment for cardiac surgery
- Radio-frequency ablation forceps/pen for cardiac surgery
- Surgical navigation and positioning system (with robotic arm and end effector)
- Endoscopic surgical system
- Control system for vascular interventional surgery
- Therapeutic ventilator
- Home healthcare environment ventilator
- External defibrillation equipment
- Haemodialysis equipment
- Continuous blood purification equipment
- Artificial liver device
- Implantable circulation assistance equipment
- Implantable drug infusion equipment
- Syringe pump (Class III)
- Needle-free injectors
- Infusion pump (Class III)
- Insulin pump (Class III)
Table: Product catalogue for high use‑risk products – according to the NMPA Guideline (as of October 2024)
3Scope of Usability Activities and Documentation
The extent of usability engineering activities expected by the NMPA primarily depends on the following aspects:
The use‑risk level of the medical device
The availability of comparable equivalent medical devices that are already available on the Chinese market
The availability of human–machine interaction information for the identified equivalent medical devices
Depending on these variables, the NMPA expects the submission of one of the following two documents:
- Use error evaluation report
- Usability engineering research report
The following flowchart illustrates the recommended approach for determining the extent of usability engineering effort.
In the following, the individual steps are discussed in detail.
Step 1: Determination of the use‑risk level of the medical device
As an initial step, the use‑risk level of the product must be determined. In this context, the NMPA follows the established approach of both the FDA and IEC 62366‑1 by emphasizing that, due to the typically limited availability of data for risk classification, the focus should be placed on the severity of harm rather than on the probability of occurrence. In particular, the NMPA follows the approach of critical tasks, as already established by the FDA. In this regard, the NMPA defines the following:
- Incorrect use of Class II medical devices (medium use‑risk) may result in minor injuries.
- Incorrect use of Class III medical devices (high use‑risk) may result in serious injury or death. This concept is comparable to the FDA’s critical task approach.
According to the NMPA, the following characteristics are considered indicators of a high use‑risk product:
- Completely new ways of use
- Long learning curves
- Use by lay users
- High operational complexity
In addition, the use‑risk level of a medical device can also be determined based on post‑market surveillance data and recalls:
The assessment is substantively based on ISO 14971:2019. The outcome is a determination of whether a medical device is classified as having a high use‑risk level or not.
Step 2: Equivalent medical device comparative evaluation on the Chinese market If the medical device under development is not classified as a product with a high use‑risk level, the usability engineering process may be shortened and a use error evaluation report can be prepared directly.
However, if a product with a High Use‑Risk level is present, an equivalent medical device comparative evaluation on the Chinese market must be conducted. For this purpose, a systematic search shall be performed to determine whether equivalent medical devices are already available on the Chinese market. Relevant sources include, in particular, NMPA registrations and publicly available product information.
Define clear criteria to justify equivalence. These may, for example, include the following aspects:
- alignment with respect to the intended use,
- alignment with respect to intended user profiles,
- alignment with respect to functional principles, and
- alignment with respect to characteristic user interface (UI) features (controls, displays, and labels),
in combination with equivalent user tasks and workflows.
The search shall be documented in a traceable manner, including the search strategy, sources consulted, identified products, and the rationale for inclusion or exclusion. If an equivalent medical device exists in China, human–machine interaction data related to this device should be specifically collected and compiled, for example in the form of an equivalent medical device comparative evaluation report, which may form part of the usability engineering research report.
Step 3: Summative usability evaluation If no equivalent medical device can be demonstrated, validation is to be conducted in the form of a summative usability test. This may be carried out either in‑house or by an external usability testing service provider (agency or test laboratory). However, the NMPA clearly emphasizes that the individuals involved in conducting the evaluation must possess expertise in usability testing of medical products and devices, and that the evaluation must not be conducted by individuals who are or were part of the development team. The summative usability evaluation is to be designed as a validation study.
For this purpose, representative Chinese users shall be recruited in accordance with the intended user profiles. Similar to the FDA, the NMPA recommends conducting the usability test with 15 participants per relevant user group. With regard to the conduct of the usability test, realistic simulation of the use environment shall be ensured in the sense of a simulated use test. In this context, labels, instructions for use, and any required product training shall also be considered in the intended market configuration.
Attention should also be paid to any potentially required training decay. In line with other standards and guidelines, use errors, close calls, and use difficulties shall be recorded and analyzed with regard to their root causes. In cooperation with risk management, this serves to generate a sufficient data basis to demonstrate the effectiveness of the risk control measures and to support the acceptability of the residual risk. If the data are sufficient, the usability engineering research report shall be compiled.
4How are the Use Error Evaluation Report and the Usability Engineering Research Report structured?
Depending on the specific case, either the use error evaluation report or the usability engineering research report provides the complete line of argumentation and body of evidence demonstrating the safe and effective use of the medical device for the NMPA. The following table presents the recommendations contained in the Guideline regarding the structure of the two documents:
Use Error Evaluation Report
- Basic information
- Level of risk of use
- Core elements
4a. Analysis of post-market use problems of similar medical devices
5a. Use-risk management
6a. Conclusion
Usability Engineering Research Report
- Basic information
- Level of risk of use
- Core elements
4b. Usability engineering process
5b. User interface requirements specification
6b. Use-risk management
7b. Verification and validation of the user interface
8b. User interface traceability analysis
9b. User training scheme
10b. Conclusion
In the following, the individual chapters of the two documents are explained in greater detail (use error evaluation report (a), usability engineering research report (b)).
Chapter 1: Basic information
Both documents begin with the basic information. The content is comparable to that of the use specification and primarily relates to general product descriptions. However, user profiles and the description of the use environment are to be provided in Chapter 3, “Core elements”.
Chapter 2: Level of risk of use
In the chapter
level of risk of use, the derivation of the use‑risk level for the product under consideration shall be presented clearly and comprehensibly. Compared to
IEC 62366‑1 and the
FDA Guidance, this chapter is new and goes beyond the typical documentation contained in the usability engineering file. While a risk‑based approach is also applied in IEC 62366‑1:2015, for example with regard to the selection of hazard‑related use scenarios or the definition of critical tasks in accordance with FDA Guidance, a more detailed justification of the risk assessment is required here.
Chapter 3: Core elements
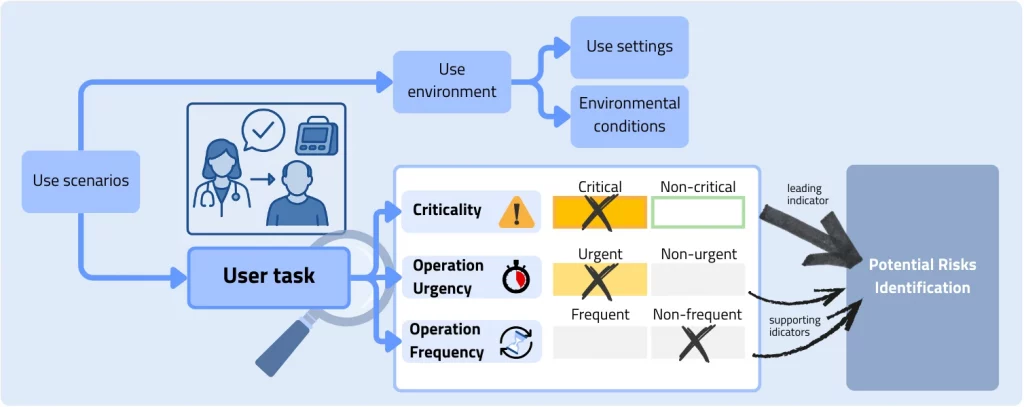
In the chapter core elements, the NMPA requires the creation of use scenarios understood as a sequence of human–machine interactions. In addition to the requirements of the IEC standard, these should also include textual and/or pictorial references to the user interface. The use scenarios should furthermore contain detailed descriptions of the use settings and environmental conditions. A key extension compared to IEC 62366‑1 is the classification of the considered user tasks along three central dimensions: Criticality, operation urgency, and operation frequency. This detailed categorization supports the systematic identification of potential risks within the framework of an NMPA‑compliant task analysis.
Practical examples for the general illustration of the classification of different user tasks
Infusion pump
Setting the flow rate
User task classification:
- critical,
- non-urgent,
- non-frequent
Criticality – Setting the flow rate determines the amount of medication or fluid administered. An incorrectly set value may result in over‑infusion or under‑infusion and can immediately cause critical physiological conditions (e.g., hypotension, hyperkalemia, shock, respiratory depression – depending on the active substance). The task is therefore critical.
Operation urgency – The setting is typically not performed under time pressure. It is carried out prior to the start of therapy or during planned adjustments. Although the potential harm is high, there is usually no second‑ or minute‑level time pressure during input. The task is therefore non‑urgent.
Operation frequency – Setting or adjusting the flow rate is not required on a continuous basis. It is an episodic action performed at the start of the infusion or during individual therapy adjustments. The task is therefore non‑frequent.
Ventilator
Immediate increase of oxygen concentration
User task classification:
- critical,
- urgent,
- non-frequent
Criticality – The oxygen concentration has an immediate effect on the patient’s oxygenation. An insufficient increase of the O₂ fraction can rapidly lead to hypoxia, which may cause acute organ damage or circulatory failure. The task is therefore critical.
Operation urgency – The need to increase oxygen delivery often arises suddenly, for example in the event of oxygen saturation decline, respiratory insufficiency, or unexpected clinical deterioration. In such situations, a response is required within seconds. The task is therefore urgent.
Operation frequency – An immediate increase in oxygen delivery is not performed routinely. It occurs only in the presence of clinical complications or unstable patient conditions. The task is therefore non‑frequent.
User task classification:
- non-critical,
- non-urgent,
- frequent
Criticality – Confirmation of an alarm does not directly affect the patient’s physiological parameters. It serves system interaction rather than direct therapy adjustment. Potential harm arises only indirectly, for example through alarm fatigue or missed critical alarms. The task is therefore non‑critical.
Operation urgency – Confirmation of an alarm is not time‑critical, as the clinically relevant action (e.g., intervention in a critical condition) must be performed independently of alarm confirmation. The confirmation primarily serves alarm management rather than immediate patient stabilization. The task is therefore non‑urgent.
Operation frequency – Confirmation of alarms occurs very frequently in clinical environments, particularly in intensive care units. The high alarm rate results in regular interaction with the alarm interface. The task is therefore frequent.
Chapter 4a : Analysis of post-market use problems of similar medical devices
This analysis report may be prepared as part of a clinical literature search and typically includes information on the search object (the medical device), the search content (how the search was conducted and which sources were reviewed), as well as the search results (including the preparation of a document list and the submission of full texts). As a general rule, the search should cover the past five years. It should be noted that the scope of the literature search should include both the major global databases for medical device adverse events and recalls, as well as Chinese and international literature databases.
Chapter 4b : Usability engineering process
In this chapter, the NMPA requires the illustration of the project‑specific usability engineering process by means of a flowchart. A table of contents for the usability engineering file shall also be provided, along with a brief description of the content and requirements of each activity performed within the process.
Chapter 5a & 6b: Use-risk management
The NMPA Guideline requires the submission of risk management documentation for the medical device to be registered, including the clear identification of the use‑related risks contained therein. Alternatively, a separate use‑related risk management file may be submitted. The use‑related risk management should include an analysis of use errors of similar medical devices that are already known based on post‑market surveillance data. In addition, it should cover a comprehensive risk analysis of all known use errors of the submitted medical device as well as the corresponding risk control measures, in order to demonstrate that the remaining use‑related residual risk is acceptable.
Chapter 5b: User interface requirements specification
This section requires the presentation of the user interface requirements specifications of the medical device, which is comparable to Chapter 5.6 of IEC 62366‑1:2015/AMD1:2020. If no separate user interface requirements specification file is available, the product requirement specification file may be submitted, provided that the user interface specification contained therein is clearly identified.
Chapter 7b: Verification and validation of the user interface
Here, the NMPA requires a description of the content and requirements of the evaluation activities related to the verification and validation of the user interface of the medical device. The term “verification” is largely equated with formative evaluation, while “validation” is equated with summative evaluation. In comparison with other standards and guidelines, it should be emphasized that the NMPA Guideline requires the submission of a user interface verification, which essentially corresponds to the formative user interface evaluation of the medical device. If a comparative evaluation with an equivalent medical device is conducted, this replaces the plan and report of the summative usability test.
Chapter 8b: User interface traceability analysis
At this point, the creation and submission of a user interface traceability analysis report is required, which transparently and comprehensibly demonstrates the relationships between the user interface requirements, the design, verification (formative evaluations), validation (summative evaluation), and risk management.
Chapter 8b, “User interface traceability analysis”, represents a novel requirement compared to other commonly used usability standards and guidelines. We recommend the development of a matrix structure that presents and cross‑references all previously described activities. This should be complemented by detailed descriptions of the relationships between the individual activities, explaining how the activities interact and interrelate.
Chapter 9b: User training scheme
If applicable, a training scheme for the medical device to be registered shall be developed at this point, describing the user training scheme, the materials, methods, and trainers used, and providing evidence of the evaluation of training effectiveness.
Chapter 6a & 10b: Conclusion
Here, the overall usability engineering process shall be briefly described in conclusion, essentially in the form of a management summary. In particular, it shall be demonstrated to what extent the use‑related residual risks have been reduced to an acceptable level, and that the safety and effectiveness of the user interface are ensured.
5When are usability tests required to be conducted in China?
Whether summative usability tests with the medical device are required is determined by the decision logic described in Chapter 3, “Scope of usability activities and documentation,” with the use‑risk level and the available data basis regarding equivalent medical devices being decisive. For products with high use‑risk and without robust evidence of an equivalent medical device, the NMPA generally expects a summative usability evaluation in the form of simulated use tests with Chinese users in a sufficiently simulated Chinese use context / simulation environment. The Guideline requires at least 15 Chinese participants per relevant user group in order to reliably identify use errors.
The Guideline provides for careful selection of participants, training where applicable, and data collection with a focus on the critical tasks. The Guideline emphasizes that individuals who frequently participate in usability tests of the same device or other devices from the same manufacturer should be excluded. Notably, compared to other guidelines, the NMPA Guideline explicitly expects a justification if no device training is required for test participants. The test reports should contain detailed information on the objectives, simulation conditions, results with respect to use errors and the associated root causes, as well as any deviations.
Once it has been determined that usability tests are required to be conducted in China, the question of practical implementation arises. While the NMPA does not specify a concrete qualification profile, it formulates clear requirements regarding the qualification and independence of the individuals conducting the tests – the evaluation shall be planned, conducted, and analyzed by individuals with demonstrable experience in usability testing of medical devices. Members of the manufacturing company who are heavily involved in the development of the device should not be assigned to conduct the evaluation.
For international manufacturers, two implementation models have essentially become established in practice:
- China‑specialized usability testing laboratories Conducting the study through partners with experienced usability testing personnel and their own infrastructure (laboratories, recruitment panels), with close substantive coordination with the manufacturer’s central usability team.
- Manufacturer‑internal implementation via local subsidiaries
Conducting the study at a Chinese subsidiary or in a company‑owned usability laboratory, provided that the moderating personnel are independent of the development team and have sufficient experience in usability testing; often in combination with external consulting to ensure NMPA‑compliant documentation.
For the Chinese authority, it is less decisive whether the study is conducted in a manufacturer’s laboratory, at an external test laboratory, or in a hospital, but rather whether the design, conduct, and documentation of the evaluation transparently demonstrate that all critical tasks can be performed safely under Chinese conditions. Clear documentation of the roles (who plans, moderates, and analyzes), the qualifications of the involved parties, and the interfaces to risk management is therefore an essential component of the documentation to be submitted.
6Conclusion – How to successfully prepare usability documentation for registration on the Chinese market
The “Guideline for Registration Review of Usability Engineering of Medical Devices” issued by the NMPA is the central reference for the assessment of usability within the Chinese registration procedure. For manufacturers with an established usability engineering process in accordance with IEC 62366‑1:2015, this means that the fundamental workflow—use specification, hazard-related use scenarios, formative and summative evaluations—remains familiar. However, the NMPA shifts the focus from a primarily internal usability engineering file toward reviewable documents that must stand on their own within the registration dossier (use error evaluation report or usability engineering research report). The recommended structure of these documents presented in Chapter 4 precisely reflects this dossier‑oriented approach.
For successful registration in China, it is therefore not sufficient to simply “submit” an existing IEC 62366‑1 process. What is decisive is that manufacturers consistently translate their usability evidence into the NMPA framework: with a clearly defined use‑risk level, a transparent decision logic (use error evaluation report vs. usability engineering research report), China‑specific summative usability tests where applicable, and reviewable documentation within the registration dossier. Manufacturers that proceed in such a systematic manner not only increase their chances of a timely market approval.
They also ensure that their products are used safely and effectively by the appropriate user groups in everyday clinical practice in China, in accordance with the labeling – thereby positioning themselves sustainably in one of the world’s most important MedTech markets.