Tailoring the Usability Engineering Process according to IEC 62366-1
How much effort is actually required?
April 2025
Executive Summary
This article examines how the usability engineering effort for medical devices can be appropriately adjusted to the specific situation without losing sight of the regulatory requirements. The international standard IEC 62366‑1:2015 specifies the requirements for the usability engineering process within the development of medical devices. Its focus is on reducing use errors that may result from inadequate usability and that could ultimately pose a risk to patients, users, or third parties. The standard is deliberately structured as a process‑oriented framework in order to accommodate different device types, risk levels, and development scopes. For this reason, the standard provides a certain degree of flexibility to tailor the extent of the usability engineering activities and adapt them to the respective development project.
The tailoring of the usability engineering effort is an integral and necessary aspect of applying IEC 62366‑1 to medical devices. It enables manufacturers to adapt the usability engineering process to the specific characteristics of the medical device, particularly its user interface, and its intended use. By considering factors such as the prevailing complexity of the user interface or the severity of potential harm related to use, a manufacturer can derive arguments to implement a tailored, efficient, and still comprehensively safe usability engineering process. How this can be carried out in practice is explained in detail in the present technical article.
1Objective of tailoring the usability engineering effort
Tailoring (adapting the extent of activities) serves the objective of conducting the usability engineering process in relation to the type of device, the risk level, and the use environment. Activities that are necessary for identifying and mitigating potential use errors must primarily be carried out in full depth. This allows the effort for documentation, analysis, and evaluation to be scaled efficiently without sacrificing essential insights.
By tailoring the usability engineering effort, the manufacturer gains the ability to customize certain activities. At the same time, the manufacturer remains responsible for ensuring that the existing safety requirements are met throughout medical device development. When implementing such a process adjustment, it is therefore essential to understand why the respective adaptation is permissible. Each adaptation should be supported with appropriate rationale and documented accordingly. The overarching objective remains the same: the development of safe and effective medical devices.

2The basis for tailoring
Because medical devices and their user interfaces can vary significantly in terms of complexity, use environment, and potential risks, it is not always necessary or practical to apply the usability engineering process to the same extent for every device. IEC 62366‑1:2015 acknowledges this and permits tailoring of the usability engineering effort. The rationale behind this lies in the need to design the process flexibly so that it can be adapted to the specific characteristics of the medical device and its intended use. The regulatory basis for this is provided in Clause 4.3, “Tailoring of the usability engineering effort,” of IEC 62366‑1:2015, which explicitly addresses this possibility for adaptation.
3Which factors influence the tailoring and extent of the usability engineering activities?
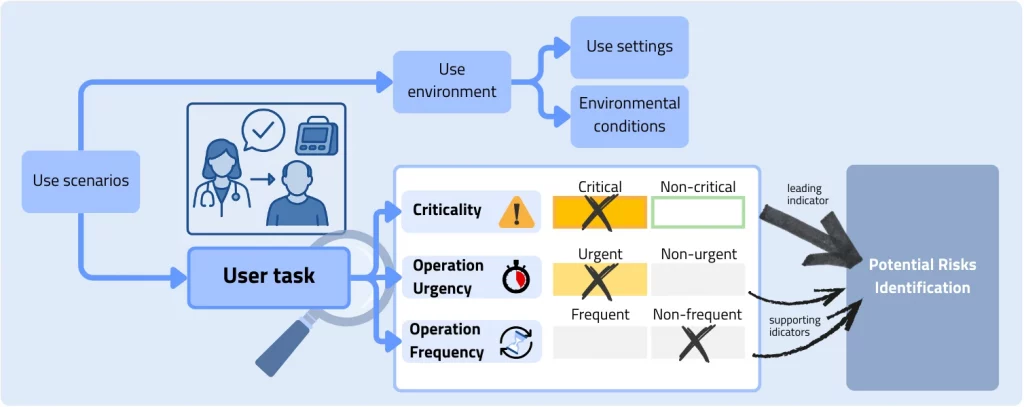
In Clause 4.3 of IEC 62366‑1:2015, the standard identifies several factors that may be used to justify tailoring the usability engineering process effort within a given medical device development project. Both the level of effort and the selection of methods and tools used to carry out the usability engineering process can be adapted based on the following factors:
Extent and complexity of the user interface: The standard states that the extent and complexity of the user interface can serve as a basis for justifying the tailoring of the usability engineering effort. A more complex user interface typically requires a higher usability engineering effort to identify and mitigate relevant use errors than a simpler one. The terms “extent” and “complexity” can be interpreted in various ways and may include aspects such as heterogeneity or the number of interaction elements.
Severity of harm associated with the use of the medical device: This is based on the rationale that devices for which use errors may lead to severe harm should undergo a more intensive usability engineering process to minimize risks than devices with a potentially lower severity of harm. The primary inputs for this assessment are the severity definitions of the company’s internal risk matrix and the detailed analysis of the hazard‑related use scenarios.
Extent or complexity of the use specification: A broad or extensive variation in intended uses, users, or use environments indicates the need for a high usability engineering process effort, whereas a simple use specification with only one user group and a clearly defined use environment may justify a lower level of effort.
Presence of a user interface of unknown provenance (UOUP): If a medical device has a user interface of unknown provenance in accordance with the UOUP definition in IEC 62366‑1:2015 (Annex C), the significantly reduced usability engineering process described therein may be applied to reduce effort. Put simply, a UOUP refers to a user interface from an already developed medical device for which no sufficient records of the usability engineering process according to the current standard are available. However, it is essential to examine the situation carefully and verify that the conditions defined in the standard for applying the UOUP process are fully met before proceeding along this path.
Extent of modification to an existing user interface of a medical device that has already undergone the usability engineering process: For minor modifications to medical devices that have already been cleared in accordance with the usability engineering process, the usability engineering effort can be focused on the modified user interface elements and their impact on the use of the device. If the modifications do not affect the user interface or the Use Specification, no additional usability engineering effort may be necessary. The key question often is: Does the modification introduce new potential use errors that could lead to risks in a non‑acceptable region?
After the fundamental factors that can be used as a rationale for tailoring the usability engineering process have been outlined, the following section illustrates their application using practical examples.
4Practical application examples
To illustrate the previously outlined theoretical factors in practice, the following section compares two medical devices with very different user interfaces and use environments.

As an example of a medical device with a user interface that might justify a low usability engineering effort, a lancet for blood glucose measurement is used. Possible lines of argumentation may include the following aspects:
- Low‑complexity user interface: It could be argued that the user interface of this manually operated lancing device for capillary blood sampling in individuals with diabetes typically consists of a mechanical release button and a simple depth adjustment via a rotating mechanism. In addition, the device contains no software components. Consequently, the human‑device interaction is limited to a few clearly defined steps.
- Low assumed severity of harm: The analysis of identified hazard‑related use scenarios may indicate a low to medium severity of harm (e.g., minor injury without the need for medical treatment and a low risk of infection).
- Low complexity of the use specification: It could be argued that the user profiles can be clearly defined and that users are generally familiar with the device.
In contrast, the assessment of an integrated anesthesia workstation with ventilation, monitoring, gas supply, and touchscreen control may lead to the conclusion that extensive usability engineering activities are necessary. This conclusion is based on the following points:
- High‑complexity user interface: The medical device includes a wide range of user interface elements. These may include multiple display layers with touchscreen control, an alarm system, as well as numerical input functions.
- High assumed severity of harm: The analysis of the hazard‑related use scenarios indicates severity levels in the high to critical range (e.g., related to lack of ventilation during induction of anesthesia or delays in life‑saving interventions) as a consequence of potential use errors.
- High complexity of the use specification: The operation of this medical device is likely carried out by multiple user groups. The Use Specification may therefore include several intended user profiles, such as anesthesiologists, nursing staff, or technicians, which in turn may require configurable, highly individualized user profiles.
The brief comparison of these medical devices illustrates how differently the individual factors may be assessed. This, in turn, provides corresponding flexibility for tailoring the extent of the usability engineering effort.
5How should one proceed, and which activities can be tailored?
The standard and the associated technical report IEC TR 62366‑2:2016 indicate usability engineering process activities that must be carried out, whereas other activities allow for greater flexibility. In this context, we recommend the following approach.
Step 1 – Establish the decision basis
As mentioned at the beginning, safe medical device development is the central focus of the standard. To ensure this, the following essential foundational activities must first be carried out:
- The conceptual description of the user interface (user interface description) of the medical device, which includes the safety‑related user interface characteristics
- The creation of a use specification
The execution of a use‑related risk analysis by means of:
- Analysis of the available post‑production / post‑market information (with a focus on known use errors that may lead to use‑related risks)
- Analysis of all hazard‑related use scenarios (with a focus on potential use errors that may lead to use‑related risks)
Ideally, this information is based on extensive user research activities as well as a well‑established internal data foundation regarding complaints and claims. This should be supplemented by a thorough review of relevant databases. In addition, the systematic identification of potential use errors as part of the development of the hazard‑related use scenarios is required.

Step 2 – Analyze the influencing factors
If comprehensive knowledge in the form of the user interface description, the use specification, and the use‑related risk analysis is available, a detailed analysis of the previously mentioned influencing factors can be carried out. This includes, for example, examining the complexity of the user interface and the Use Specification, as well as the assumed severity of harm.
Step 3 – Define and document the number and extent of the further usability engineering activities
Depending on the completeness and level of detail of the Use Specification as well as the known and potential use errors, the extent of the subsequent usability engineering activities is now defined. This includes in particular the further extent of
- User research activities: The extent depends on the magnitude of existing gaps in the use specification, such as with respect to user groups, prevailing workflows, or use environment specifications.
- User interface design iterations: Depending on the complexity, a few design cycles may be sufficient, or extensive iterative development cycles may be required. Design iterations should always be planned hand in hand with formative evaluations.
- Formative evaluations: These are used to identify problems associated with the use of still‑immature user interfaces in order to conduct design optimizations based on the findings. The number and depth of these evaluations may vary and can be influenced to some extent (for example, through the choice of evaluation methods or the number of participating test users) and are often dependent on the extent and maturity of the available user interface prototypes.
- Summative evaluation activity: This serves to provide objective evidence that the use‑related residual risk is at an acceptable level. The method most commonly applied here is the usability test. The scope of a summative evaluation is determined by the risks and the diversity of the hazard‑related use scenarios, as well as the associated user groups. If modifications to the user interface are required after the summative evaluation, additional evaluations may become necessary if the changes could introduce further use‑related risks. However, these can be focused on the affected interactions or interface elements.
Important – Keep the deliverable in mind: In particular, the definition of the number and extent of the formative usability evaluations to be conducted, as well as the methodology of the summative evaluation, should be documented in the user interface evaluation plan.
Conclusion
IEC 62366‑1:2015 enables a flexible and risk‑based approach to applying the usability engineering process. This is both reasonable and necessary to achieve an optimal balance of safety, effort, and product quality. Tailoring the usability engineering effort allows manufacturers to structure this process efficiently by adjusting its extent to the specific risks and the complexity of the product. Strategic tailoring—meaning a customized definition of the usability engineering activities—makes it possible to align regulatory compliance with economic efficiency within the development process. Take the time early in your development project to establish clarity in this regard, as it will save you significant effort and difficulties as the project progresses.
However, when tailoring the process, always ensure the consistency of your rationale in order to demonstrate compliance with the requirements of IEC 62366‑1:2015. A final remark: IEC 62366‑1 focuses strictly on applying the usability engineering process to optimize the usability of medical devices with respect to safety. More extensive usability activities may therefore not be regulatory requirements and may not be necessary to “get through” the process. However, such additional activities often lead to the user‑centered improvements that ultimately determine user satisfaction in daily practice and, consequently, the success or failure of your product.
Disclaimer
The information on standards and guidelines presented in this technical article has been prepared to the best of the author’s knowledge and expertise. It reflects solely the opinion of the author. No guarantee can be given with regard to the completeness, timeliness, or accuracy of the information provided. Standards and guidelines are subject to regular revisions and amendments, which may not always be immediately reflected here. This article does not constitute binding advice and does not replace a review of the applicable standards and guidelines by qualified experts or official bodies. For the application and interpretation of standards and guidelines, the currently valid original documents and the responsible organizations are authoritative.

As usability engineering specialists, we at USE‑Ing. are pleased to support you in tailoring the usability engineering effort as part of your medical device development. Do you have any questions? Feel free to contact us.